AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Bohrs model of atom3/24/2024  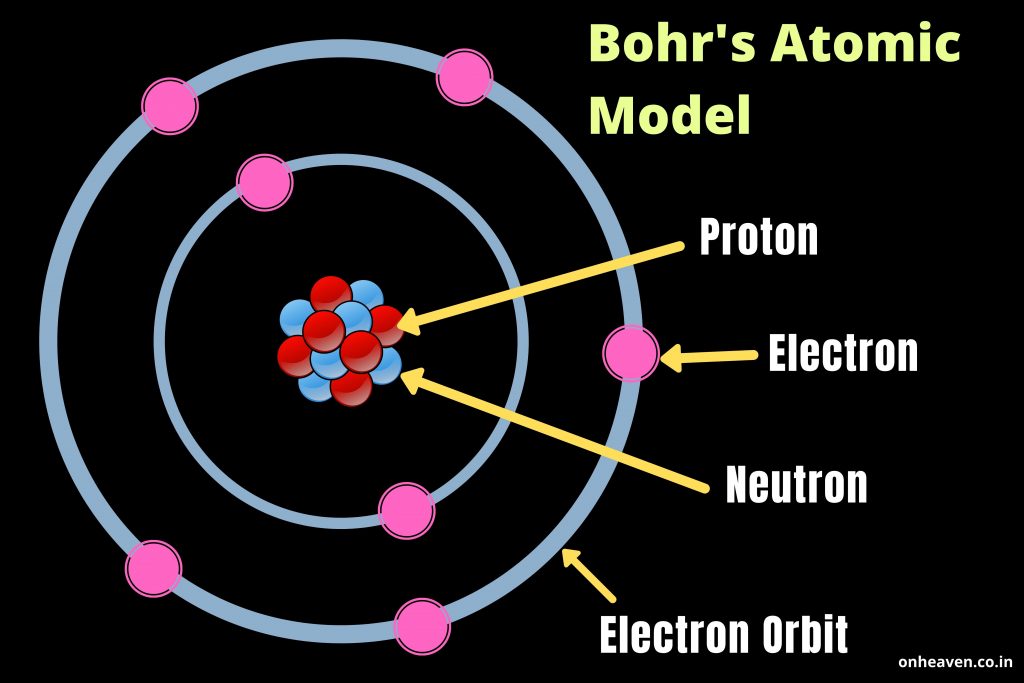
Understanding Bohr's model requires some knowledge of electromagnetic radiation (or light).īohr's key idea in his model of the atom is that electrons occupy definite orbitals that require the electron to have a specific amount of energy. The Bohr model was the first atomic model incorporating some quantum mechanics. Thus, the electron in a hydrogen atom usually moves in the n 1 orbit, the orbit in which it has the lowest energy. One of the fundamental laws of physics is that matter is most stable with the lowest possible energy. In 1913, the Danish physicist Niels Bohr proposed a model of the electron cloud of an atom in which electrons orbit the nucleus and were able to produce atomic spectra. 1 k hc( 1 n2 1 1 n2 2) The lowest few energy levels are shown in Figure 6.2.1. The existence of the atomic spectra is support for Bohrs model of the atom. These difficulties cast a shadow on the planetary model and indicated that, eventually, it would have to be replaced. Bohrs model suggests that the atomic spectra of atoms is produced by electrons gaining energy from some source, jumping up to a higher energy level, then immediately dropping back to a lower energy level and emitting the energy difference between the two energy levels. Equation 7.4.12 gives the energies of the electronic states of the hydrogen atom. Hence, the energy of the electron in an atom also is quantized. Furthermore, Rutherford's model was unable to describe how electrons give off light forming each element's unique atomic spectrum. and this simplifies the allowed energies predicted by the Bohr model (Equation 7.4.11) as. If the electron circling the nucleus in an atom loses energy, it would necessarily have to move closer to the nucleus as it loses energy, and would eventually crash into the nucleus. Bohr built upon Rutherfords model of the atom. This is, after all, how we produce TV signals. Bohr thought that electrons orbited the nucleus in circular paths whereas in the modern view atomic electron structure is more like 3D standing waves. It was already known that when a charged particle (such as an electron) moves in a curved path, it gives off some form of light and loses energy in doing so. Bohr proposed that electrons do not radiate energy as they orbit the nucleus, but exist. The electron is not allowed to occupy any of the spaces in between the orbits. Unfortunately, there was a serious flaw in the planetary model. According to the Bohr model, or planetary model, the electrons encircle the nucleus of the atom in specific allowable paths called orbits. \): Niels Bohr with Albert Einstein at Paul Ehrenfest's home in Leiden (December 1925). 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
